√70以上 homogeneous and heterogeneous definition and examples 316941-What is the meaning of homogeneous and heterogeneous
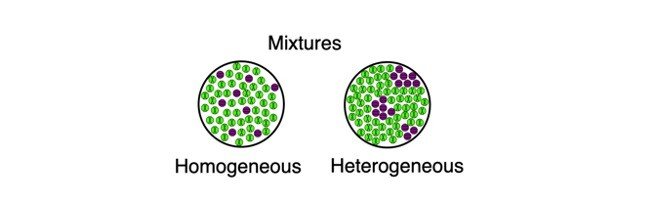
The difference between heterogeneous and homogeneous mixtures is the degree to which the materials are mixed together and the uniformity of their composition A homogeneous mixture is a mixture in which the components that make up the mixture are uniformly distributed throughout the mixture The composition of the mixture is the sameThat is, it is a set of ordered pairs ( x , y ) consistingHeterogeneous solids, liquids, and gases may be made homogeneous by melting, stirring, or by allowing time to pass for diffusion to distribute the molecules evenly For example, adding dye to water will create a heterogeneous solution at first, but will become homogeneous over time
What Is Homogeneous Welding Examples Advantages Disadvantages
What is the meaning of homogeneous and heterogeneous
What is the meaning of homogeneous and heterogeneous-When two or more elements or compounds mix together, not necessarily in a definite ratio and do not interact chemically, then the resulting substance is knowA homogeneous equilibrium is one in which all of the reactants and products are present in a single solution (by definition, a homogeneous mixture ) Reactions between solutes in liquid solutions belong to one type of homogeneous equilibria The chemical species involved can be molecules, ions, or a mixture of both For the homogenous reaction




Examples Of Heterogeneous Mixtures Types Made Simple
A complete answer to the question What are Examples of homogeneous mixtures and heterogeneous mixtures?Examples of Homogeneous Mixtures Solid, Liquid and Gas A chemical mixture combines two substances that maintain their own properties when combined Heterogeneous mixtures are made up of a nonuniform composition, while homogeneous mixtures are made up of a uniform composition For example, water and sand is a heterogeneous mixture — you can• Apple juice is homogeneous • Orange juice with pulp is heterogeneous • Chocolate dough is homogeneous • Italian salad dressing is heterogeneous
A phase is any part of a sample that has a uniform composition and properties By definition, a pure substance or a homogeneous mixture consists of a single phase A heterogeneous mixture consists of two or more phases When oil and water are combined, they do not mix evenly, but instead form two separate layersElement and Compound are examples of pure substances Mixtures are made up of two or more substances which are the constituents of mixture The constituents of mixture can be in any ratio Mixtures can be divided into 1 Homogeneous mixtures 2 Heterogeneous mixtures Homogeneous mixtures have uniform composition For instance, sugar in water, water in alcoholIn this animated lecture, I will teach you the concept of mixture, different types of mixture, homogeneous mixture, heterogeneous mixture, difference between
For example, the definition of an equivalence relation requires it to be symmetric Listed here are additional properties that a homogeneous relation may satisfy In mathematics , a binary relation over sets X and Y is a subset of the Cartesian product X × Y {\displaystyle X\times Y} ; Examples of heterogeneous mixtures include sand, oil and water, and chicken noodle soup What is solid solid heterogeneous mixture? There are two types of mixtures, homogeneous mixtures and heterogeneous mixtures Vinegar is an example of a homogeneous mixture of acetic acid and water Heterogeneous means "different throughout" A heterogeneous mixture has large parts that are different from each other



The Definition And Measurement Of Heterogeneity Translational Psychiatry



Heterogeneity And Heterogeneous Data In Statistics Statistics How To
Examples of a heterogeneous mixture Colloid A colloid is an example of a heterogeneous mixture where the components exist in two distinct phases; A mixture is any substance that consists of two or more components, and it can be either homogeneous or heterogeneous Homogeneous mixtures contain two or more components that cannot be visually distinguished or separated into the individual components Examples of homogeneous mixtures include Salty water — a mixture of salt and Mixture of Oil and Water is a heterogeneous Mixture We are able to see oil and water clearly separately in the mixture Example 2 Mixture of Salt (Sodium Chloride) and Iron filings is a heterogeneous mixture The particles of salt and Iron filings can be seen and distinguished easily Difference between Homogeneous and Heterogeneous mixtures




Homogeneous Mixture Definition Examples Tutors Com



Difference Between Homogeneous And Heterogeneous Mixtures Definition Composition Characteristics Examples
The components in a heterogeneous mixture can easily be seen with a naked eye, but for better results, you can utilize a microscope Examples Homogeneous Mixture Some examples of homogeneous mixtures are most alloys, seawater, brass, vinegar, air, blood, natural gas, etc Heterogeneous Mixture Salt and pepper together make a heterogeneousThe components can be visibly distinguished Heterogeneous Definition and Examples Biology Online Dictionary Heterogeneous in the largest biology dictionary onlineThis chemistry video tutorial explains the difference between homogeneous and heterogeneous mixtures within the subtopic of the classification of matter It



Heterogeneity What Is It And Why Does It Matter Students 4 Best Evidence



Homogeneous Vs Heterogeneous Energy Education
Heterogeneous mixtures, on the other hand, don't showcase similar physical properties Water with salt, Chlorine dissolved in water, Vinegar dissolved in water, Seawater, Sugar water, etc, are examples of homogeneous mixtures, whereas Soups and broths, Cereal with milk, Oil and water, Water and sand, Refreshments, etc are examples of Heterogeneous Mixtures Heterogenous mixtures have a mixed composition which may vary from point to point Visibility of Components Homogeneous Mixtures Components are not visible to the naked eye Heterogeneous Mixtures Components can be seen easily and distinguished Phase Separation Homogeneous Mixtures The whole mixture is in the same phaseHaving widely unlike elements or constituents opposed to homogeneous The attraction between the different kinds of electricity and magnetism



Homogeneous Mixtures Examples Definition And Types




26 Homogeneous And Heterogeneous Catalysis Chapter
Homogeneous and heterogeneous mixtures In chemistry, if the volume of a homogeneous suspension is divided in half, the same amount of material is suspended in both halves of the substance An example of a homogeneous mixture is air In physical chemistry and materials science this refers to substances and mixtures which are in a single phase Homogeneous Consisting of or composed of similar elements or ingredients, of a uniform quality throughout Origin gr Genos = kind Homogeneous Definition and Examples Biology Online DictionaryAns Following are the 10 different examples of homogeneous mixtures 1) Salt and water solution 2) Vinegar 3) Alcohol and water 4) Air 5) Sea water 6) Coffee 7) Carbonated drinks 8) Sugar and




Examples Of Homogeneous Mixtures Solid Liquid And Gas




Examples Of Heterogeneous Mixtures Types Made Simple
Solid heterogeneous mixtures can contain liquid or gaseous components, but as a whole, they act like solids mixed nuts – Mixed nuts at a party are a type of Homogenous (definition) generally means "of the same kind" or alike In biology, it is the old term for homologous, which is defined as "having corresponding parts, similar structures, or the same anatomical positions" Etymology from Latin homo, meaning "same" and "genous" means "kind" Variant homogeneous Antonym heterogeneousAbout heterogeneous mixture homogeneous mixture worksheet The heterogeneous mixture – homogeneous mixture worksheet with answer key is below The worksheet gives common examples of mixtures, in addition to some pure, unmixed substances Learn how to classify these examples of mixtures below
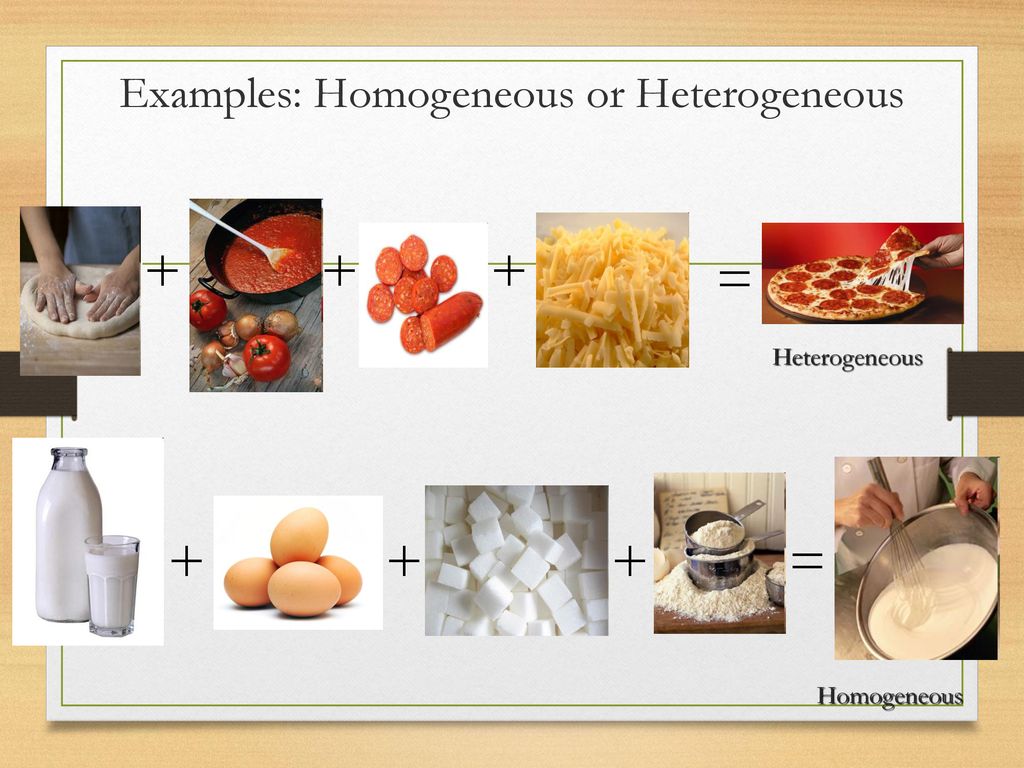



Separation Methods Ppt Download



1
Heterogeneous mixture is a mixture with a nonuniform composition When you mix two components that remain separate from each other, that mixture is called a Heterogeneous mixture Examples of Heterogeneous mixture Concrete is an example of a Heterogenous mixture A mixture of Cement and Water A mixture of cold drinks and ice cube is also an example of a Heterogeneous Heterogeneous and Homogeneous Mixtures In the lesson, you learned about heterogeneous mixtures and homogeneous mixtures Another name for a homogeneousThe whole world is a solid heterogeneous mixture!
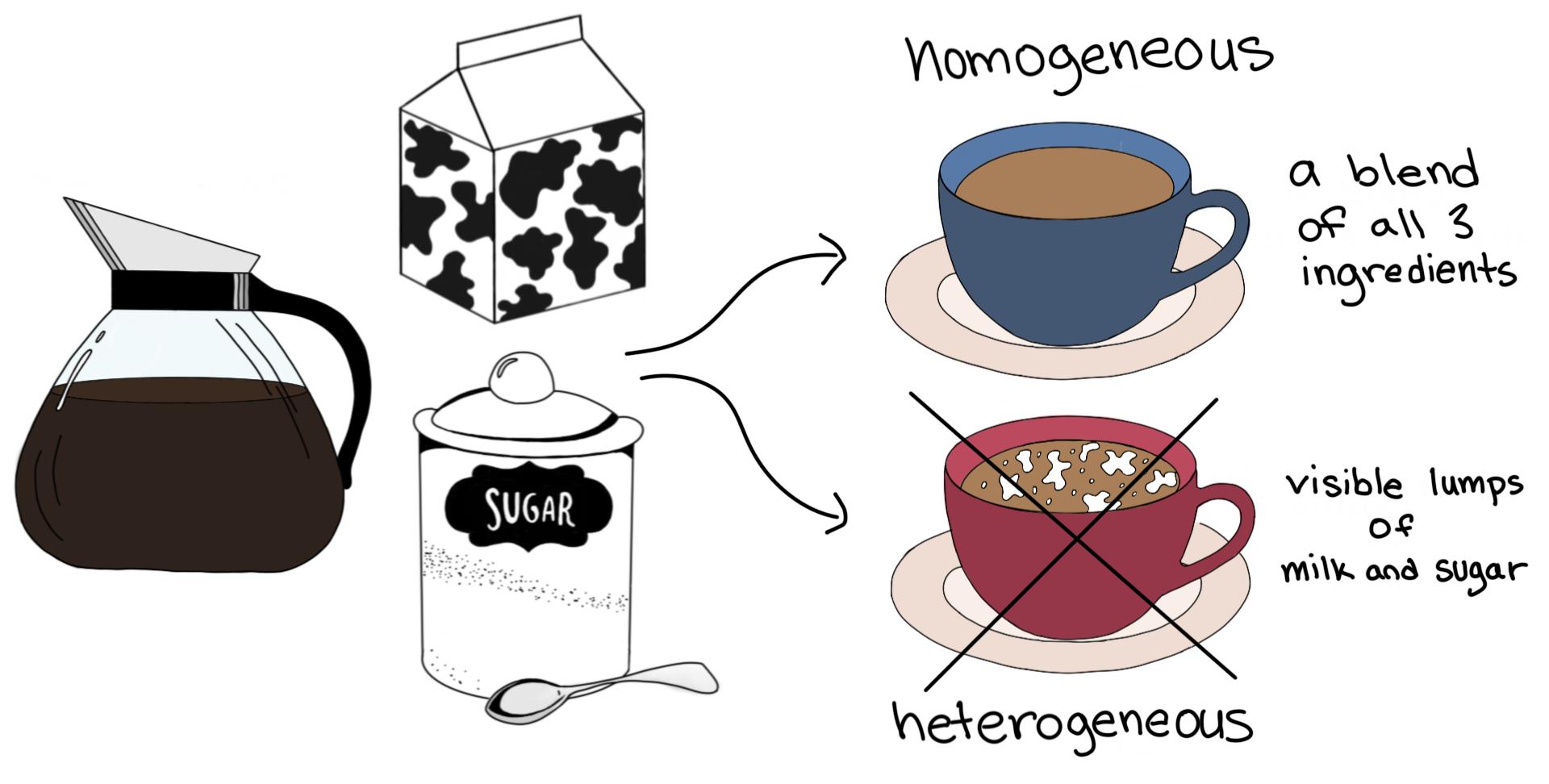



Homogeneous Vs Heterogeneous Mixtures A Comparison Expii




Homogeneous And Heterogeneous Mixtures Card Sorting Activity Heterogeneous Mixture Sorting Activities Sorting Cards
Thus, the main difference between homogeneous and isotropic is that homogeneous refers to the uniformity of structure and isotropic refers to the uniformity of physical properties Key Areas Covered 1 What is Homogeneous – Definition, Properties, Examples 2 What is Isotropic – Definition, Properties, Examples 3Moreover, a heterogeneous equilibrium example is also provided in order to learn about the difference between homogeneous and heterogeneous equilibrium The example is given below H 2 O (s) ⇌ H 2 O (l) Homogeneous Equilibrium Example A homogeneous equilibrium can further be divided into two categories In the first category, the number of Heterogeneous grouping is a type of distribution of students among various classrooms of a certain grade within a school In this method, children of approximately the same age are placed in different classrooms in order to create a relatively even distribution of students of different abilities as well as different educational and emotional needs




Homogenous And Heterogenous Mixtures Chemistry For Non Majors




Examples Of Homogeneous Mixtures And Heterogeneous Mixtures Youtube
Heterogeneous Mixture A mixture that is not uniform in its composition; The reaction between acid and metal is a heterogeneous reaction A reaction between a gas and a liquid, as between air and seawater, is heterogeneous A reaction at the surface of a catalyst is heterogeneous In contrast, a reaction between two miscible liquids or between two gases is homogeneousIn this animated lecture, I will teach you about 10 examples of homogeneous mixtures and 10 examples of heterogeneous mixtures, the meaning of homogeneous, t




Homogeneous Mixture Definition Examples Tutors Com




Homogeneous Mixture Definition Examples Video Lesson Transcript Study Com
Homogeneous Mixture Mixtures which have uniform composition throughout are called Homogeneous Mixture For example – mixture of salt and water, mixture of sugar and water, air, lemonade, soda water, etc Mixture of salt in water is an example of homogeneous mixture In this mixture, the boundary of salt and water cannot be differentiated Homogenous populations are alike and heterogeneous populations are unalike Homogenous means alike Heterogenous means unalike or distinct from one another Thus, a homogenous population has little variation You could refer to a specific trait, such as hair color or you could refer to genetic diversity For example, a population of humans that has inhabited anHeterogeneous consisting of dissimilar elements Heterogenous 1 not originating within the body;




What Do You Need To Know About Heterogeneous And Homogeneous Mixtures




Homogenous And Heterogenous Mixtures Chemistry For Non Majors
The root of "geneous" is a Greek word, "genos," meaning a group, type, or stock So, homogeneous means all the same group, and heterogeneous means all different groups together Think of two different bowls of soup tomato soup is homogeneous, while the vegetable soup is heterogeneous2 of foreign origin; A mixture of apples and oranges is heterogeneous A bucket of rocks contains a heterogeneous mixture of shapes, sizes, and composition A group of different barnyard animals is heterogeneous A mixture of oil and water is heterogeneous because the




10 Homogeneous Mixture Examples In Daily Life Studiousguy




Heterogeneous Mixture Definition Science Trends
The terms heterogeneous and homogeneous are two different mixtures that refer to chemistry when two or more substances without a chemical reaction are mixed together, a mixture is formed the mixtures are very important in chemistry there are two major types of mixtures Homogeneous and heterogeneous mixtures the basic difference between heterogeneous and homogeneousFor example, a salad Unlike homogeneous mixtures, in heterogeneous mixtures it is very easy to identify, even with the naked eye, what are the different components that make them up This makes it much easier to separate these mixes at the same time For example water and oil / water and sand Examples of Heterogeneous Mixtures Aerosols3 heterogeneous Heterogeneous, with that fourth e, is the opposite of homogeneous (which is different from homogenous)) Most of us will never have use for heterogenous in its most strictly defined senses, where it is a term used almost exclusively in




Difference Between Homogeneous And Heterogeneous Material Youtube




Homogeneous Vs Heterogeneous Mixtures A Comparison Expii
Solid homogeneous mixture brass is an alloy that is made from metal copper (Cu) and zinc (Zn) Liquid homogeneous mixture a saline solution that is the mixture of water and salt Gas homogenous mixture air is a mixture of different gases such as oxygen, Definition and Examples Examples of Heterogeneous Mixtures Concrete is a heterogeneous mixture of an aggregate cement, and water Sugar and Homogeneous Vs Heterogeneous Mixtures In a homogeneous mixture, the components are present in the same proportion no Homogenization A heterogeneousIncongruous Composed of parts of different kinds;




What Is The Difference Between Heterogeneous Mixture Vs Homogenous Mixture Brainly Com



3
By definition, a pure substance or a homogeneous mixture consists of a single phase A heterogeneous mixture consists of two or more phases When oil and water are combined, they do not mix evenly, but instead form two separate layers Each of the layers is From the practical point of view, homogeneous welding is always easier compared to heterogeneous welding due to inherent chemical compatibility among filler and base metals Various similarities and differences between homogeneous welding and heterogeneous welding are discussed in the following sectionsDispersed phase and continuous phase The insoluble particles of a colloid do not settle down completely as the particles are small in size, usually ranging between 107 to 103 cm




Natural And Man Made Examples Of Homogeneous Mixture Science Struck




Homogeneous And Hetrogeneous Mixtures Definition Examples Teachoo
The homogenous mixture/solution has only one phase that is solid, liquid and gas Examples for the same are; For example, when you add some mud or sand in the water, you form a heterogeneous mixture Q What are 10 examples of homogeneous mixtures?Homogeneous mixtures are also called as solutions Uniform composition Example rainwater, vinegar etc Heterogeneous mixture This is a type of mixture in which all the components are completely mixed and all the particles can be seen under a microscope We can easily identify the components and more than one phase can be seen by naked eyes




Homogeneous Mixture Example Food
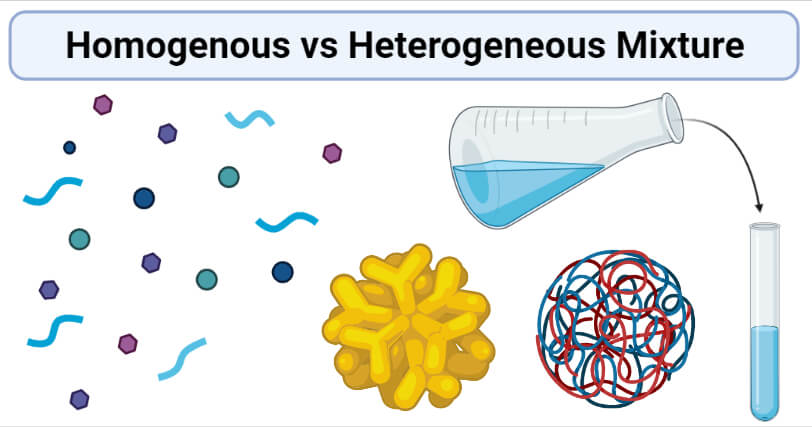



Homogenous Vs Heterogeneous Mixture Definition 8 Key Differences Examples




Definition Of Homogeneous And Heterogenous Systems In This Review Download Scientific Diagram



1




Mixtures And Solutions Cpd Rsc Education




Heterogeneous Mixture Homogeneous Mixture Worksheet Easy Hard Science




What Do You Need To Know About Heterogeneous And Homogeneous Mixtures



Homogeneous And Heterogeneous Mixture Nine Science
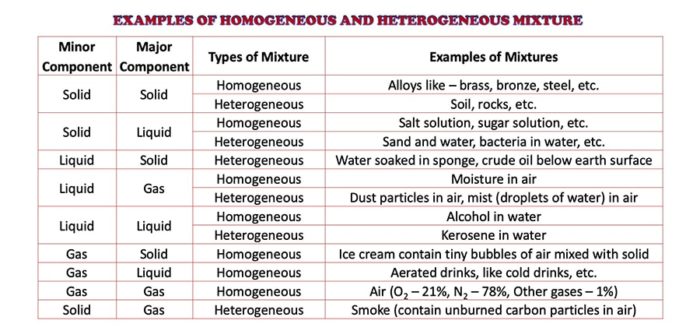



Homogeneous Heterogeneous Mixture Definition Examples Selftution



Mixture



Difference Between Homogeneous And Heterogeneous Welding




Heterogeneous Mixture And Homogeneous Mixture Youtube




Heterogeneous Mixture Homogeneous Mixture Worksheet Easy Hard Science



Difference Between Homogeneous And Heterogeneous Mixtures Definition Composition Characteristics Examples
/definition-of-heterogeneous-mixture-and-examples-605206_final23-ecfa4da6517640429448462eae1f09f7.png)



Definition Of Heterogeneous Mixture With Examples



Heterogeneous Mixture Lesson For Kids Definition Examples Video Lesson Transcript Study Com




فصل 1 Br Section A Br Some Basic Definitions




Heterogeneous Products Definition Overview Video Lesson Transcript Study Com




Matter Anything With Mass And Volume Ppt Video Online Download




Difference Between Homogeneous And Heterogeneous Mixtures Definition Composition Characteristics Exam Heterogeneous Mixture Chemistry Basics Biology Facts
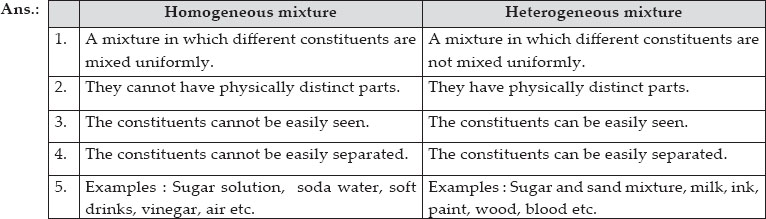



Ncert Solutions Is Matter Around Us Pure Chemistry Class 9




Heterogeneous And Homogeneous Mixture Differences Videos Examples




Heterogeneous Vs Homogeneous Mixtures
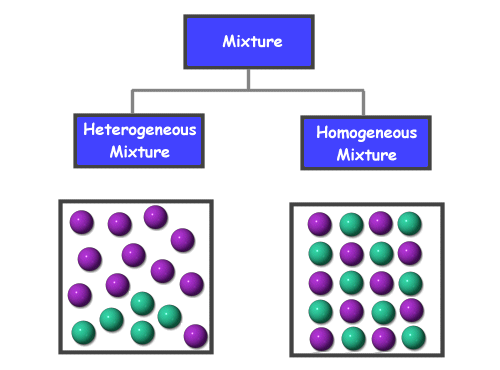



Chemistry For Kids Chemical Mixtures




Homogeneous And Heterogeneous Mixture Difference Between Homogeneous And Heterogeneous Mixture Youtube




What Is A Homogeneous Mixture Definition And Examples




Homogeneous And Heterogeneous Mixtures Examples Classification Of Matter Chemistry Youtube
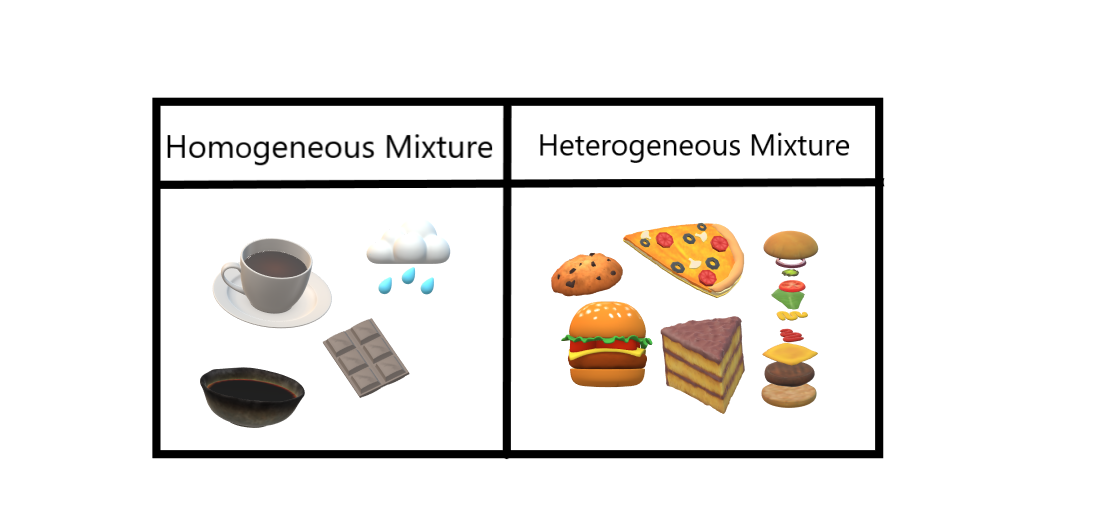



Homogeneous And Heterogeneous Mixtures Geeksforgeeks



What Is Homogeneous Welding Examples Advantages Disadvantages



Mixture
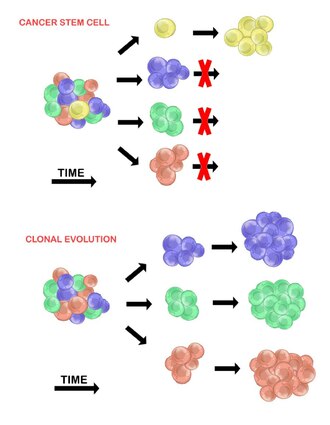



Tumour Heterogeneity Wikipedia




Difference Between Homogeneous And Heterogeneous Homogeneous Vs Heterogeneous
/TC_606106-heterogeneous-and-homogeneous-mixtures1-5ac4f1a9642dca0036847e52.png)



Heterogeneous Vs Homogeneous Mixtures




Homogeneous Mixture Definition Examples Tutors Com




Heterogeneous Mixture Definition Science Trends




Lesson Explainer Mixtures Nagwa
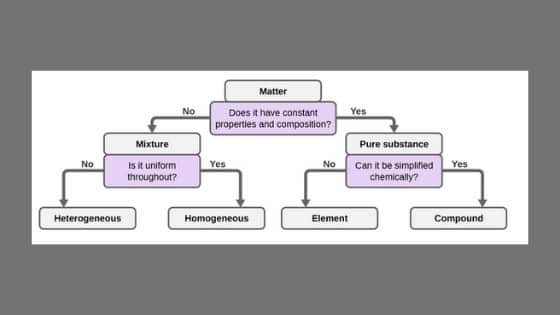



Homogeneous Mixture And Heterogeneous Mixture Ncert Books




What Is A Heterogeneous Mixture Definition And Examples
/TC_606106-heterogeneous-and-homogeneous-mixtures1-5ac4f1a9642dca0036847e52.png)



Heterogeneous Vs Homogeneous Mixtures




Homogeneous Mixture Examples In Nature
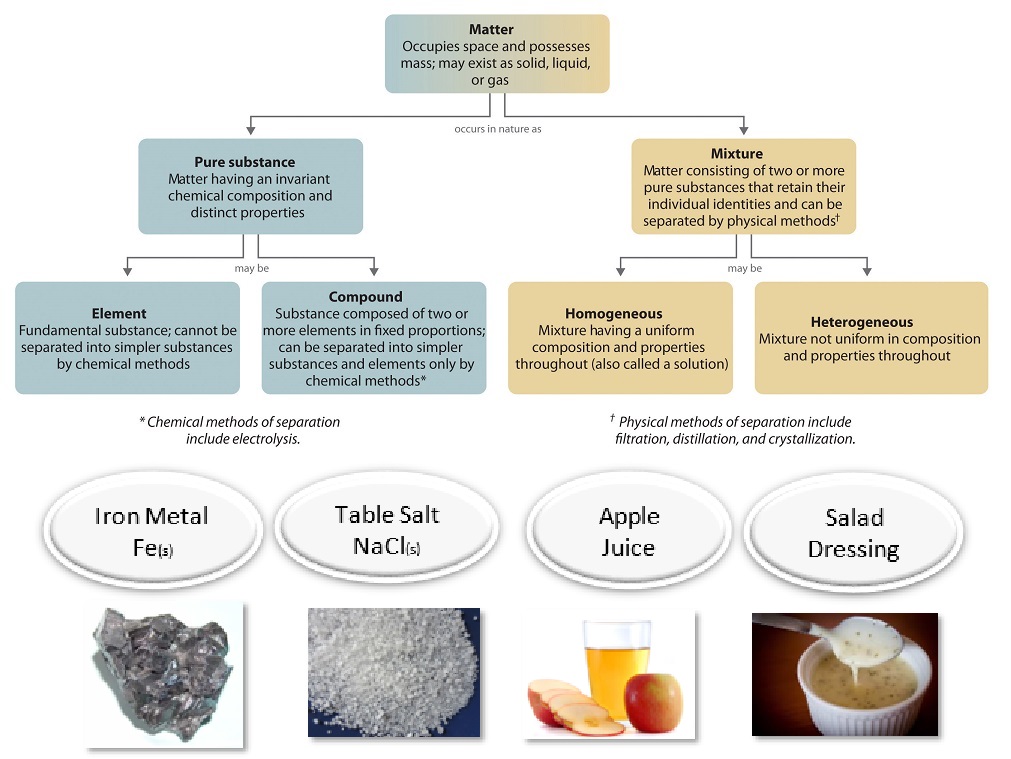



3 4 Classifying Matter According To Its Composition Chemistry Libretexts



Mixture




Homogeneous Heterogeneous Mixture Definition Examples Selftution




Homogeneous Vs Heterogeneous Mixtures A Comparison Expii
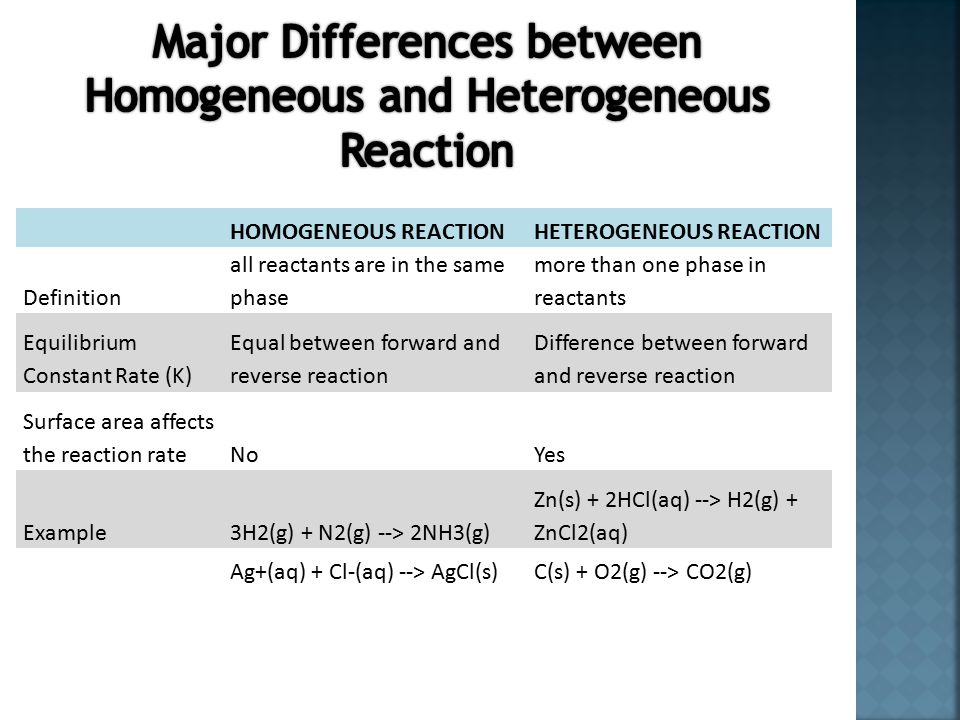



What Is The Difference Between Homogeneous And Heterogeneous Equilibrium Slide Share




Elements Compounds And Mixtures Worksheet Chemistryrack




Homogenous Vs Homogeneous What S The Difference Writing Explained




Differentiate Between Homogeneous And Heterogeneous Mixtures With Examples




Ways To Separate Mixtures Definition Types Homogeneous Heterogeneous Mixture Eschool




Difference Between Homogeneous Mixture And Heterogeneous Mixture




Homogenous Definition And Examples Biology Online Dictionary




Q2 Differentiate Between Homog Lido
/GettyImages-182154729-56a133dd5f9b58b7d0bcfe8b.jpg)



Homogeneous Definition And Examples



Homogeneous Heterogeneous Mixture Definition Examples Selftution
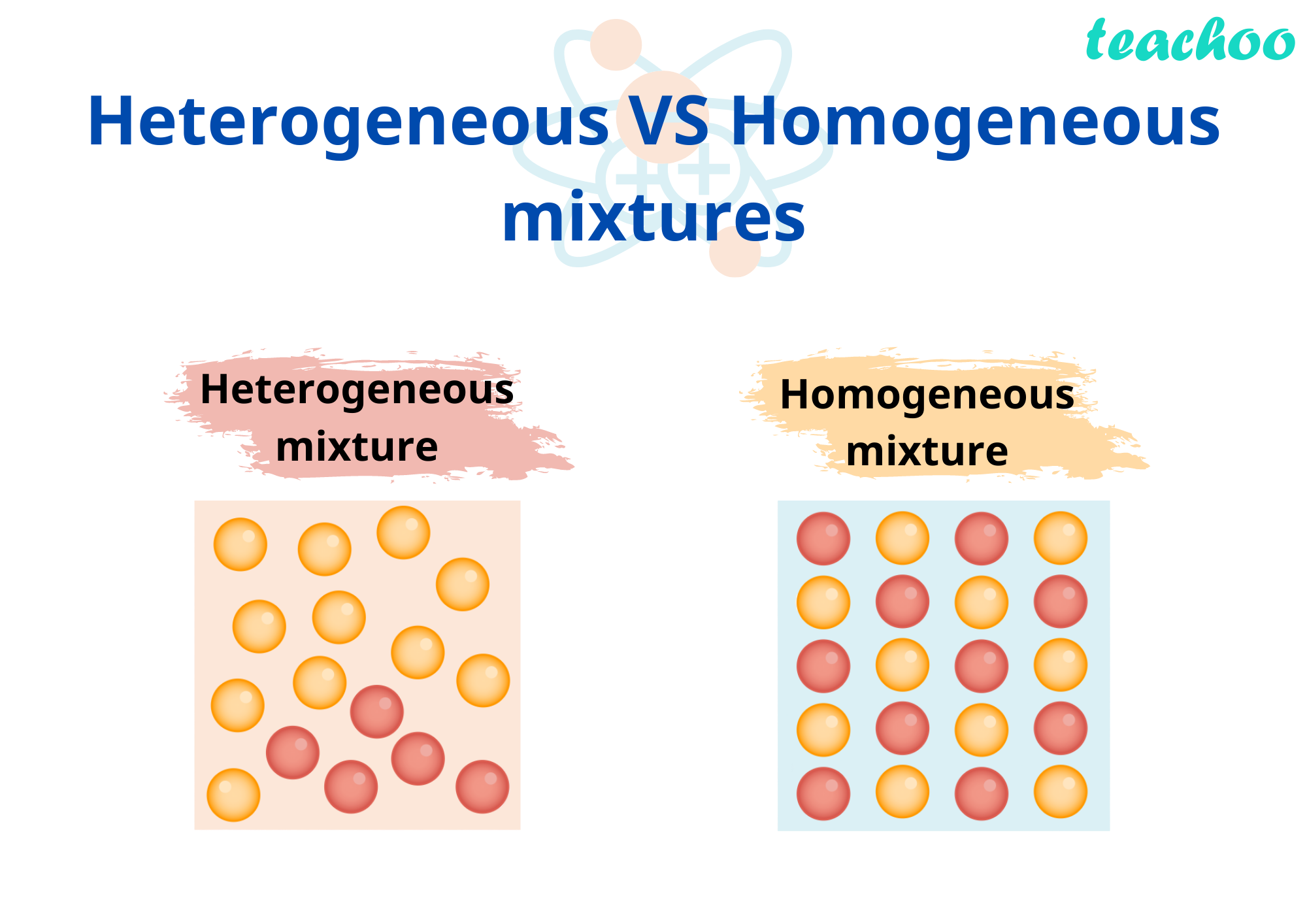



Homogeneous And Hetrogeneous Mixtures Definition Examples Teachoo




Matter Anything With Mass And Volume Ppt Video Online Download
/GettyImages-548326197-58fe30b63df78ca159cb3f67.jpg)



Heterogeneous Definition Science




Heterogeneous Mixture Definition Examples Video Lesson Transcript Study Com




Homogeneous Mixture Examples Found At Home




Homogenous And Heterogenous Mixtures Chemistry For Non Majors




Homogenous Definition And Examples Biology Online Dictionary




5 Examples Of Homogeneous Mixture For Chemistry Class Science Trends




Homogeneous And Heterogenous Mixture Definition Examples Diagrams




Homogeneous Mixture Examples Found At Home




What Is The Difference Between Homogeneous And Heterogeneous Mixture
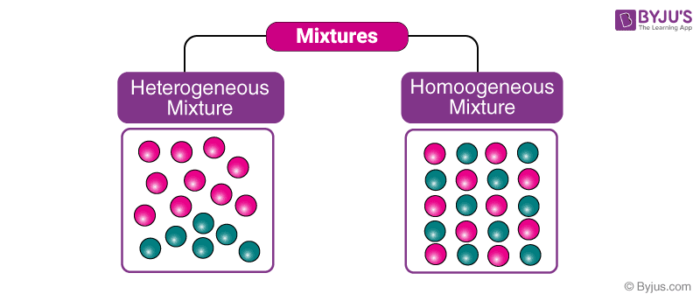



Heterogeneous And Homogeneous Mixture Differences Videos Examples




Classification Of Matter Adapted By J Stevens From Ppt Video Online Download




Homogeneous Vs Heterogeneous Mixtures A Comparison Expii




Homogeneity And Heterogeneity Wikipedia




Mixtures Homogeneous And Heterogeneous Mixtures Ppt Video Online Download




Homogeneous Mixture Science Struck




What Is A Homogeneous Mixture Definition And Examples




10 Examples Of Mixtures




Homogeneous And Heterogeneous Mixtures Card Sorting Activity Heterogeneous Mixture Sorting Activities Matter Worksheets



1




Homogeneous Vs Heterogeneous Mixtures A Comparison Expii
コメント
コメントを投稿